Treatment of Neuropathic Pain With Venlafaxine a Systematic Review
Introduction
Depression and chronic pain are disabling and often concomitant pathologies; both are currently ii of the principal public wellness problems (1, 2). Major depressive disorder (MDD) is the most prevalent psychiatric affliction and has been recognized as a critical target of intervention in the psychiatric field (3, 4). All the same, depression remains underdiagnosed and consequently, undertreated (4, 5). Furthermore, generalized anxiety disorder (GAD), obsessive-compulsive disorder (OCD), and postal service-traumatic stress disorder (PTSD) are common psychiatric comorbidities with MDD, normally lead to worse prognosis and compromise the remission of MDD symptoms (6).
GAD is one of the well-nigh prevalent psychiatric disorders, affecting half-dozen% of the population during their lifetime (7, 8). GAD is a chronic condition that severely affects the quality of life, due to its repercussion on working and social functioning (viii). Even though anxiety is a widespread symptom, the diagnosis of GAD requires a complex procedure of screening for a right diagnosis (9). Moreover, GAD is usually associated with other clinical conditions such as MDD or pain syndromes, affecting drastically prognosis, and treatment efficacy (10).
Chronic pain is a persistent pain condition with a dual dimension, based on the signaling machinery pathways: nociceptive and neuropathic pain (NP) (11). Specifically, NP has a strong emotional implication, and has been associated with worse quality of life, and clinically, it is related with affective disturbances such every bit depression, anhedonia, working retention dysfunction, sleep disturbances, anxiety, and impaired cognition (12–14). Moreover, chronic pain involves a stress component that might play a bidirectional predictive part. That is, chronic stressful events produce biochemical and pathophysiologic alterations that lead to stress-related mood disorders, that besides may increase the risk of chronic pain or exacerbate it (14, 15).
On the other hand, fibromyalgia syndrome (FMS) is a chronic widespread pain condition with high heterogeneity clinically and etiologically (16, 17). It is estimated that 4–6% of adults worldwide suffer from FMS, whose incidence is increased in women (18). The well-nigh debilitating symptom of FMS is generalized pain. Other symptoms such as fatigue, sleep disturbances, cognitive impairment, or headache are as well part of the cadre symptoms of FMS (19). Concomitantly, MDD symptoms also overlap with the FMS, as well as GAD that is significantly higher in patients with FMS (20, 21).
There is a possible connexion betwixt anxiety, depression, and stress urinary incontinence (SUI). Evidence suggests that both feet and depression are predictor of SUI onset (22). SUI is characterized by an unintentional urinary leakage due to coughing, exertion or sneezing, which increment the intra-abdominal and bladder pressure that overcome urethral resistance (23). Serotonin (5-HT) pathways are involved in this disorder. Thus, 5-HT induces the urethral sphincter closure past inhibition of the micturition reflex (22).
In this perspective, duloxetine is a potential handling for these dissimilar clinical atmospheric condition, but with shared pathophysiological pathways. Duloxetine is a serotonin-norepinephrine reuptake inhibitor (SNRIs) canonical as a first-line drug to treat MDD, GAD, diabetic peripheral neuropathy (DPN), FMS and SUI (24–28). As a SNRIs, duloxetine increases both levels of serotonin and norepinephrine which are directly correlated with adverse events, such equally tachycardia, hypertension, among others (29). Pharmacokinetic and pharmacodynamic data of duloxetine accept been reported for several studies, whose evidence suggests that duloxetine is generally well-tolerated (30–33). Thus, the main goal of this systematic review was to determine the efficacy, tolerability, and safety of duloxetine in the treatment of the clinical conditions for which it is canonical.
Methodology
Study Blueprint
A qualitative systematic review of literature was performed, post-obit the 2009 Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines, and the Joanna Briggs Establish (JBI) critical appraisal checklist for the dissimilar types of studies reviewed (34). This systematic review aimed to depict and synthetize the evidence and potential benefits of duloxetine.
Protocol Registration
The protocol was registered in the international database PROSPERO of the National Institute for Health and Enquiry (NIHR) with the code CRD42020153634.
Eligibility Criteria
To accomplish this comprehensive systematic review the following inclusion criteria were causeless: all studies written in English and focused on human adults (at least xviii years old) with MDD, GAD, NP, FMS, or SUI (clinical atmospheric condition for which duloxetine has approval), published until 01/09/2020. Those studies whose primary outcomes were efficacy, tolerability, and/or rubber of duloxetine were included. Studies focused on other psychiatric or neurological status such as Parkinson'due south illness, Alzheimer's disease, chronic non-neuropathic hurting, bipolar, schizoaffective, and schizophrenia disorders were excluded. Moreover, qualitative research reports were besides excluded, as well every bit reports whose analyses were based on pooled integrative data analysis of randomized command trials (RCTs). Eligible designs included RCTs, non-randomized experimental studies, case-command, and cohort studies, which outcomes were quantitatively measured past social, functional, cognitive, quality of life (QoL), or treatment emergent adverse events (TEAEs) instruments.
Data Sources and Search Strategy
Studies were selected from PubMed, Medline, Web of Scientific discipline, and PsycINFO electronic databases, introducing the search terms: "duloxetine" AND "major depressive disorder" OR "MDD"; "duloxetine" AND "generalized anxiety disorder" OR "GAD"; "duloxetine" AND "neuropathic pain"; "duloxetine" AND "fibromyalgia"; and "duloxetine" AND "stress urinary incontinence." Ii contained researchers (DRA and JMO) conducted the search strategy, applying the filters described in the inclusion criteria to refine the process and obtain concise results.
Report Selection
Authors independently screened the reports. Firstly, titles and abstracts were reviewed to evaluate their concordance with our requirements. Secondly, the total-text of the potential studies were screened and appreciated and those that met our inclusion criteria were selected. Finally, 85 studies were included in this systematic review. Discrepancies were resolved through discussion amid the authors until consensus was reached.
Information Extraction and Synthesis
To summarize the relevant information of the selected studies, the authors extracted and performed a Tabular array with the post-obit data: offset writer and year of publication, number of participants, gender, mean historic period (years), duloxetine dose per day (mg), duration of the treatment (weeks), diagnosis scales or other clinical measuring instruments, relevant statistical results, type of study, and the principal outcomes. The process of synthesis allowed a disquisitional appraisal of the studies and the event size calculation based on the statistical information reported by studies.
Results
Search Results
The first stage of the searching process comprised a search in the electronic databases using specific search terms, where 2,661 reports were identified. In the second stage, inclusion criteria were practical, duplicate reports were removed, and 727 records by title and abstruse were studied. Three hundred and forty-2 studies were analyzed in the third stage, and their full-text versions were carefully examined. In this stage, they were 85 eligible studies that met the inclusion criteria and the JBI recommendations (Supplementary Tables 1–4). Finally, in the 4th stage, studies on the different clinical conditions for which duloxetine treatment is approved −32 studies on major depressive disorder (MDD), 11 studies on GAD, nineteen studies on NP, 9 studies on fibromyalgia, and xiv studies on SUI were selected (Effigy ane).
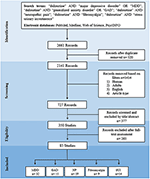
Figure 1. PRISMA 2009 catamenia diagram of search process. MDD, major depressive disorder; GAD, generalized feet disorder; NP, neuropathic pain; SUI, stress urinary incontinence.
Study Characteristics
The eligible studies were examined and categorized by clinical status. 5 clinical conditions were considered for the treatment with duloxetine: MDD, GAD, NP, FMS, and SUI. Thus, eighty-5 studies were scrutinized and a total of 34,808 participants were enrolled (25,448 female person, ix,108 male; and 581 participants that in their report were not differentiated past gender), with an historic period range of xviii–97 years. The studies included 21,406 patients that were treated with duloxetine with a dose ranged from 20 to 120 mg and a treatment elapsing of 12 ± 14.39 weeks. The main reasons of dropout were adverse events (59.5%), lack of effectiveness (20.iii%), patient's decision (9.5%), loss of follow-upwards (5.4%), non-adherence to handling (two.7%), hospitalizations (1.iii%), and others (1.iii%). Within adverse events, the near mutual were nausea (xviii.xiii%), dry mouth (ix.69%), constipation (7.42%), and somnolence (five.94%) (Figure two). Cardiovascular disease was an exclusion benchmark of 7% of the studies, and cardiovascular adverse events (hypertension, tachycardia, myocardial ischemia, increased claret pressure, and arrythmia) were evaluated in 68.2% of the studies, where 49.four% reported statistical insignificance for these TEAEs (P <0.05), and 11.eight% showed a correlation betwixt elevated heart rate and duloxetine treatment. Regarding the type of studies, 58.7% of the studies are RCTs, 25.nine% are cohort studies, 11.8% are quasi-experimental studies (non-randomized) and 3.v% are instance-control studies.
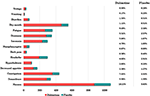
Figure two. Adverse events of duloxetine-treated patients and placebo patients. Bar graph of the most common adverse furnishings vs. the number (Northward) of participants who developed them (Ntotal duloxetine-treated patients = 4,848 and Ntotal placebo patients = three,536). The table shows the corresponding percentage.
Major Depressive Disorder
MDD studies comprised 1,836 patients that were treated with 20–120 mg of duloxetine during eight ± 17.05 weeks. This condition was diagnosed based on the Diagnosis and Statistical Manual of Mental Disorders (DSM) from their third to fifth edition. Efficacy of duloxetine was measured in 78.1% of the studies using the Hamilton Depression Rating Scale (HAMD), the Geriatric Depression Scale (GDS), the Montgomery and Asberg Depression Rating Scale (MADRS) or the Brief Pain Inventory (BPI) when pain, and MDD were concomitant. Safety of treatment with duloxetine was assessed in 25% of the studies based on TEAEs, and tolerability was evaluated in 31.3% of studies (35–66). Twenty-six studies were able to demonstrate the superiority of duloxetine over placebo or other antidepressants such as sertraline, fluvoxamine, venlafaxine, paroxetine, escitalopram, fluoxetine, and bupropion. Five studies did non discover statistical significance (P ≥ 0.05) regarding the correlation betwixt duloxetine and the outcomes and 1 report did not obtain significant results when comparing duloxetine with sertraline. Safe and tolerability were evaluated past TEAEs and the most common adverse events (AEs) were nausea, somnolence, dry mouth, hyperhidrosis, constipation, and sedation; patient's dropout rate was ~10% (Effigy ii). This result was not meaning in nearly of studies, concluding that duloxetine was safety and well-tolerated (meet Table 1).

Table 1. Characteristics of the selected studies and included in the systematic review.
Generalized Feet Disorder
11 studies focused on GAD were included in this systematic review, which involved two,608 patients treated with 20–120 mg of duloxetine with an average elapsing of treatment of 10 ± 6.59 weeks. All studies used the DSM to achieve the diagnosis. Clinical evidence was based on the correlation betwixt the Hamilton Anxiety Rating Calibration (HAMA) and the outcomes. Therefore, all studies found statistical significance (P ≤ 0.05) when measured the efficacy (90.9% of the studies), safety and tolerability (both 27.3% of the studies) (67–77). TEAEs were measured and nausea, dry mouth, dizziness, and somnolence were reported as the most frequent AEs (Figure ii). One written report reported suicidal ideation, although no statistical significance was plant between duloxetine and placebo groups (69). Duloxetine was more effective, safe and tolerated than placebo or other antidepressants (escitalopram and venlafaxine) (Table ane).
Neuropathic Pain
The selected NP studies reported the use of duloxetine in the handling of peripheral neuropathy induced by chemotherapy, diabetic peripheral neuropathic hurting (DPNP), radiculopathy and neuropathic pain associated with multiple sclerosis (NP-MS) (Tabular array 1). NP condition was diagnosed using specific criteria of pain detection, being the BPI the well-nigh commonly applied musical instrument. TEAEs were the measure for prophylactic and tolerability. The dose of duloxetine applied was ranged from 20 to 120 mg during 12 ± 14.53 weeks. The core of the studies focused on 4,627 patients with NP, where the efficacy, prophylactic, and tolerability of duloxetine were compared with placebo, anticonvulsant treatments (pregabalin and gabapentin), other antidepressants (venlafaxine and amitriptyline), or even with dissimilar daily doses of duloxetine. Thus, 78.nine% of the studies reported efficacy outcomes, 47.4 and 21.1% of the studies described safety and tolerability of duloxetine, respectively (78–96). Iii studies did not show statistical significance regarding efficacy, safety, and tolerability of duloxetine against anticonvulsants (P ≥ 0.05). Nausea, somnolence, insomnia, constipation, and decreased appetite were the almost prevalent TEAEs (Effigy two). A minority of patients discontinued the treatment with duloxetine due to AEs (12.2%). Notwithstanding, 84.two% of the studies supported the prove of duloxetine as first-line treatment of NP conditions.
Fibromyalgia
Nine studies focusing on FMS were assessed and eligible. They involved a total of 1,918 patients. The average duration of treatment was 24 ± 13.78 weeks and the dose of duloxetine oscillated from 20 to 120 mg per day (Table 1). The BPI scale and Fibromyalgia Touch on Questionnaire (FIQ) were the instruments employed to analyse the outcomes. Near studies (77.8%) evaluated the efficacy of duloxetine, and 55.6% provided information of the treatment safety (97–104, 119). Statistically significant results were obtained in 7 studies, where duloxetine improved symptomatology, reducing the pain impact registered past BPI. In contrast, two studies reported no statistical differences relative to BPI alter boilerplate and cognitive comeback in fibromyalgia patients. The duloxetine treatment was related to ~17% of agin effects. Taking all these studies into account, duloxetine showed to be safe and effective in FMS handling.
Stress Urinary Incontinence
Fourteen studies that involved half-dozen,395 patients (99.5% female person and 0.v% male person) were assessed. Duloxetine doses were between xx and 120 mg per solar day. Treatment elapsing was 12 ± 6.72 weeks and the Incontinence Episode Frequency (IEF) and Incontinence Quality of Life (I-QoL) were the instruments to measure the outcomes. All studies evaluated efficacy of duloxetine; 71.four and 7.1% of the studies measured safety and tolerability, respectively (105–118). Treatment was discontinued in 22.ane% of patients regarding TEAEs, beingness the most common fatigue, nausea, constipation, and dry mouth (Figure ii). These AEs tend to improve and disappear with continuing duloxetine therapy. In conclusion, significant results were found in all studies, proving the efficacy, condom, and tolerability of duloxetine in the treatment of SUI.
Quality Assessment
A systematic review summarizes the evidence of the relevant literature, nevertheless, an unbiased search of studies without an explicit assess strategy could lead to a poor scientific report. The relevance and quality of the studies selected and included in this systematic review fulfilled the PRISMA recommendations (120), and JBI critical appraisal guidelines. The JBI is an evidence-based arrangement that develops strategies to conduct and perform a loftier quality systematic reviews (121). Thus, the quality decision of the studies included indicates that our inquiry minimizes the chance of selection bias. Furthermore, a proficient systematic review relies on the studies it contains. Therefore, the inclusion of RCTs and clinical trials reduce the probability of bias due to their strict methodology (122).
Discussion
In the last years, mutual pathophysiological mechanisms have been identified in depression, pain, and anxiety (11). Neuropathic pain, specifically DPNP, coexists with mental disorders, predominantly with low and feet (123). Highlighting the functional harm as a consequence of unremitting pain symptoms, neuropathy has been correlated with an increased gamble of depression (124). On the other hand, the widespread spontaneous pain is the virtually debilitating symptom of FMS, that might be a link to depression and anxiety disorders as comorbid conditions (125). Due to the similar pathophysiologic mechanisms and high occurrence of FMS and depression, these clinical conditions were considered under a common approach to affective disorders, GAD and PTSD (19). Urinary incontinence is too a severe trouble that affects 15–30% of adults over 60 years, and several studies have reported a link between urinary incontinence, anxiety and low in women (22, 126).
The core of the pathophysiology of these clinical conditions is more often than not due to the disruption of five-HT and norepinephrine (NE) pathways (xix, 22, 127). The monoaminergic hypothesis is based on a fractional or total deficit of 5-HT or NE in the key nervous system (CNS) (128, 129). Somatic symptoms such as muscle tension, neuropathic and musculoskeletal pain, fatigue, or dizziness are common in MDD and GAD among other psychiatric disorders as issue of aberrant 5-HT and NE neurotransmission (130). Regarding pain, antinociception and pronociceptive modulation occurs through 5-HT receptors, in both the central and peripheral nervous systems (131). Equally in pain, SUI involves the action of monoaminergic system. Evidence demonstrates that endogenous regulation of serotoninergic and noradrenergic machinery in the spinal cord works simultaneously to maintain the reflex of urinary continence (132).
Therefore, the pharmacological treatment of clinical weather condition with similar pathophysiology involves a global perception of coexisting disorders. In this sense, antidepressants such as duloxetine accept been considered effective in the treatment of MDD, GAD, NP, FMS, and SUI (133). Duloxetine is a serotonin-norepinephrine reuptake inhibitor, that is, a potent inhibitor of 5-HT transporter (SERT) and norepinephrine transporter (Net) (134). Due to this dual machinery, its profile seems to have a different response compared to selective five-HT reuptake inhibitors (SSRIs) (135). In vivo studies, duloxetine presented preferential inhibition of 5-HT reuptake and low affinity for hitamine-H1, alpha-i-norepinephrine, v-HT(1A, 1B, 1D), muscarinic acetylcholine, and opioid receptors (136). Clinically, duloxetine has been canonical for diverse clinical conditions, acquiring new show over the years, also being prescribed to treat other neuropathic pain conditions and chronic musculoskeletal pain (eighty, 137).
In this systematic review, we considered the efficacy, prophylactic, and tolerability of duloxetine in the handling of current canonical clinical atmospheric condition. Firstly, an individual search by clinical status was achieved based on specific inclusion criteria. This strategy allowed u.s. to observe consistent results and objectively evaluate the outcomes. Apropos efficacy, duloxetine demonstrated effectiveness in over 80% of cases. Still, some TEAEs are frequent, such as dry rima oris, somnolence, nausea, constipation or hyperhidrosis, tending to decrease in time and disappear with continuing duloxetine therapy. Cardiovascular adverse events, such as hypertension, increased heart charge per unit, myocardial ischemia, are also associated with duloxetine administration (29). Within these, simply the increase in middle rate was statistically significant, although not existence clinically relevant. In sum, duloxetine was considered in all assessed reports every bit a safe and well-tolerated treatment even in cardiovascular disease, as well as in elderly patients (29, 51, 82, 93). In this sense, our results prove that duloxetine is an option with a valid and consolidated therapeutic value. Secondly, we focused on the clinical conditions' comorbidity. The coexistence of depression, anxiety, and pain is a frequent country, too every bit, depression and SUI, and FMS and depression. Therefore, the treatment with duloxetine is largely used due to its dual mechanism that ameliorate the symptoms associated with the concomitant clinical conditions (e.k., MDD and NP). Moreover, we observed a strong link between MDD and hurting. This correlation suggests a bidirectional blueprint: MDD could be a predictor of chronic hurting which in turn might predict the persistence of MDD (138). Thirdly, although the dropout rate with duloxetine treatment reaches around twenty% in certain cases, similar rates were plant in placebo and other antidepressants or anticonvulsants treatments. Finally, some considerations should exist taken into account regarding to duloxetine prescription and titration. Alcohol, tobacco and java (caffeine) are the most widely consumed psychoactive substances worldwide (139, 140). Evidence suggests that plasmatic serum levels of duloxetine were decreased (about 15%) in smoking patients due to the induction of CYP1A2 (141). Hepatotoxicity was besides observed in patients whose alcohol consumption was significant (142). Lastly, caffeine is also metabolized past CYP1A2, like duloxetine, and this may increment duloxetine serum levels. Nonetheless, this interference needs more supporting evidence.
We performed this systematic review in order to include as much evidence as possible. In this process, nosotros analyzed a big number of studies to support our conclusions. Still, we plant some limitations. The inclusion criteria exclude reports in a linguistic communication other than English. Thus, significant studies may have been missed with this strategy. Our research protocol dismisses qualitative and pooled integrative data analysis of RCTs to avoid repeated analysis of RCTs information and qualitative findings duplication. Regarding the effect size of studies, some of them did not report sufficient statistical data to compute Hedges'southward thou (e.thou., standard deviation). All the same, we decided to include these 19 studies due to their relevance respect to the evaluated outcomes.
In conclusion, there is a substantial amount of evidence in support of efficacy, safety, and tolerability of duloxetine in the treatment of MDD, GAD, NP, FMS, and SUI. The dose range of threescore–120 mg daily demonstrated efficacy in most of the studies assessed. TEAEs were balmy to moderate, and AEs decreased or remitted with continuing duloxetine treatment. Treatment discontinuation due to both AEs and ineffectiveness of duloxetine yielded enough acceptable results to conclude that duloxetine is safe and well-tolerated. In improver, duloxetine is a monotherapy approach that might be useful to treat concomitant disorders with parallel pathophysiological pathways such as MDD and NP, which is an advantage for patients (avoiding polytherapy issues) and a successful price-effective culling.
Data Availability Argument
All datasets generated for this report are included in the commodity/Supplementary Cloth.
Author Contributions
Data analysis and the first draft of the manuscript was written by DR-A and TR-B. All authors contributed to the study design, acquired data to assay, and read and approved the concluding manuscript.
Funding
This work was financially backed by the Foundation for Science and Technology (FCT, Fundação para a Ciência e Tecnologia) within the framework of grant SFRH/BD/135623/2018 awarded to DR-A and another grant of Fundación Tatiana Pérez de Guzmanel Bueno provided to CS. Our research was further supported by the Carlos Three Wellness Institute (ISCIII, Instituto Carlos III) through grant P16/00405 and co-funding awarded by the Spanish Foundation of Rare Diseases (FEDER, Fundación Española de Enfermedades Raras) to JO.
Conflict of Involvement
JO declares paid positions, honoraria or being part of advisory boards by Angelini, AstraZeneca, Bristol-Myers, Casen Ricordati, Esteve, GSK, Janssen, Lilly, Lundbeck, Novartis, Otsuka, Pfizer and Sanofi, and grants from Ministry of Health Castilian National Institute of Heath Carlos Iii, National Substance Abuse Plan, and Galician Innovation Programs (Gain) in the final ten years.
The remaining authors declare that the enquiry was conducted in the absence of whatsoever commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would similar to thank the Galicia Sur Health Inquiry Found (Instituto de Investigación Sanitaria Galicia Sur) and the University Infirmary Complex of Vigo (Complexo Hospitalario de Vigo) for their support. In addition, the authors are especially thankful for the aid provided by the Psychiatric Nursing and Psychiatry Services of the Álvaro Cunqueiro and Nicolás Peña hospitals.
Supplementary Textile
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2020.554899/full#supplementary-material
References
2. Jacob KS. Depression: a major public health problem in need of a multi-sectoral response. Indian J Med Res. (2012) 136:537–9.
PubMed Abstruse | Google Scholar
iv. Kraus C, Kadriu B, Lanzenberger R, Zarate CA, Kasper S. Prognosis and improved outcomes in major low: a review. Transl Psychiatry. (2019) 9:127. doi: ten.1038/s41398-019-0460-3
PubMed Abstract | CrossRef Full Text | Google Scholar
6. Trivedi MH, Rush AJ, Wisniewski SR, Nierenberg AA, Warden D, Ritz L, et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. (2006) 163:28–40. doi: x.1176/appi.ajp.163.1.28
PubMed Abstract | CrossRef Full Text | Google Scholar
7. Wittchen H, Kessler RC, Beesdo KK, Krause P, Höfler M, Hoyer J. Generalized anxiety and low in primary care: prevalence, recognition, and management. J Clin Psychiatry. (2002) 63:24–34.
PubMed Abstract | Google Scholar
8. Roberge P, Normand-Lauzière F, Raymond I, Luc Thou, Tanguay-Bernard MM, Duhoux A, et al. Generalized anxiety disorder in master care: mental wellness services employ and treatment adequacy. BMC Fam Pract. (2015) 16:146. doi: 10.1186/s12875-015-0358-y
PubMed Abstract | CrossRef Full Text | Google Scholar
10. Katzman MA, Bleau P, Blier P, Chokka P, Kjernisted Thousand, Van Ameringen M, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive-compulsive disorders. BMC Psychiatry. (2014) 14:1–83. doi: 10.1186/1471-244X-14-S1-S1
PubMed Abstract | CrossRef Full Text | Google Scholar
xiii. Fiore NT, Austin PJ. Are the emergence of affective disturbances in neuropathic pain states contingent on supraspinal neuroinflammation? Brain Behav Immun. (2016) 56:397–411. doi: 10.1016/j.bbi.2016.04.012
PubMed Abstract | CrossRef Full Text | Google Scholar
15. Li MJ, Liu LY, Chen Fifty, Cai J, Wan Y, Xing GG. Chronic stress exacerbates neuropathic pain via the integration of stress-Affect-related information with nociceptive information in the central nucleus of the amygdala. Pain. (2017) 158:717–39. doi: 10.1097/j.pain.0000000000000827
PubMed Abstruse | CrossRef Full Text | Google Scholar
sixteen. Arnold LM, Choy E, Clauw DJ, Goldenberg DL, Harris RE, Helfenstein M, et al. Fibromyalgia and chronic hurting syndromes: a white paper detailing current challenges in the field. Clin J Pain. (2016) 32:737–46. doi: 10.1097/AJP.0000000000000354
PubMed Abstract | CrossRef Full Text | Google Scholar
17. Tan AC, Jaaniste T, Champion D. Chronic widespread pain and fibromyalgia syndrome: life-course risk markers in young people. Pain Res Manag. (2019) 2019:1–13. doi: ten.1155/2019/6584753
PubMed Abstract | CrossRef Full Text | Google Scholar
18. De Roa P, Paris P, Poindessous JL, Maillet O, Héron A. Subjective experiences and sensitivities in women with fibromyalgia: a quantitative and comparative report. Pain Res Manag. (2018) 2018:1–8. doi: ten.1155/2018/8269564
PubMed Abstract | CrossRef Full Text | Google Scholar
20. Thorlacius S, Stefánsson SB, Ranavaya MI, Walker R. Fibromyalgia and feet disorder. Laeknabladid. (2002) 88:815–8.
PubMed Abstract | Google Scholar
21. Cardinal TM, Antunes LC, Brietzke AP, Parizotti CS, Carvalho F, De Souza A, et al. Differential neuroplastic changes in fibromyalgia and depression indexed by upward-regulation of motor cortex inhibition and disinhibition of the descending pain organisation: an exploratory study. Front Hum Neurosci. (2019) 13:138. doi: 10.3389/fnhum.2019.00138
PubMed Abstract | CrossRef Total Text | Google Scholar
22. Felde G, Ebbesen MH, Hunskaar South. Feet and depression associated with urinary incontinence. a 10-year follow-upwardly study from the Norwegian HUNT study (EPINCONT). Neurourol Urodyn. (2017) 36:322–8. doi: 10.1002/nau.22921
PubMed Abstract | CrossRef Total Text | Google Scholar
24. Jost WH, Marsalek P. Duloxetine in the treatment of stress urinary incontinence. Ther Clin Gamble Manag. (2005) 1:259–64.
PubMed Abstruse | Google Scholar
28. Iqbal Z, Azmi S, Yadav R, Ferdousi M, Kumar M, Cuthbertson DJ, et al. Diabetic peripheral neuropathy: epidemiology, diagnosis, and pharmacotherapy. Clin Ther. (2018) twoscore:828–49. doi: 10.1016/j.clinthera.2018.04.001
PubMed Abstract | CrossRef Full Text | Google Scholar
29. Park K, Kim South, Ko YJ, Park BJ. Duloxetine and cardiovascular agin events: a systematic review and meta-analysis. J Psychiatr Res. (2020) 124:109–fourteen. doi: 10.1016/j.jpsychires.2020.02.022
PubMed Abstract | CrossRef Full Text | Google Scholar
30. Nelson JC, Pritchett YL, Martynov O, Yu JY, Mallinckrodt CH, Detke MJ. The safety and tolerability of duloxetine compared with paroxetine and placebo: a pooled analysis of 4 clinical trials. Prim Intendance Companion J Clin Psychiatry. (2006) 8:212–nine. doi: 10.4088/PCC.v08n0404
PubMed Abstruse | CrossRef Full Text | Google Scholar
31. Arnold LM. Duloxetine and other antidepressants in the handling of patients with fibromyalgia. Pain Med. (2007) 8:S63–74. doi: 10.1111/j.1526-4637.2006.00178.ten
CrossRef Total Text | Google Scholar
33. Mease PJ, Russell IJ, Kajdasz DK, Wiltse CG, Detke MJ, Wohlreich MM, et al. Long-term safety, tolerability, and efficacy of duloxetine in the treatment of fibromyalgia. Semin Arthritis Rheum. (2010) 39:454–64. doi: 10.1016/j.semarthrit.2008.11.001
PubMed Abstract | CrossRef Full Text | Google Scholar
34. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health intendance interventions: explanation and elaboration. J Clin Epidemiol. (2009) 62:e1–34. doi: ten.1016/j.jclinepi.2009.06.006
PubMed Abstract | CrossRef Full Text | Google Scholar
35. Berk Grand, Plessis AD, Birkett G, Richard D, Group LDDS. An open-characterization study of duloxetine hydrochloride, a mixed sertotonin and noradrenaline reuptake inhibitor, in patients with DSM-Iii-R major depressive disorder. Int Clin Psychopharmacol. (1997) 12:137–40. doi: 10.1097/00004850-199705000-00003
PubMed Abstract | CrossRef Full Text | Google Scholar
36. Goldstein DJ, Lu Y, Detke MJ, Wiltse C, Mallinckrodt C, Demitrack MA. Duloxetine in the treatment of low: a double-blind placebo-controlled comparing with paroxetine. J Clin Psychopharmacol. (2004) 24:389–99. doi: x.1097/01.jcp.0000132448.65972.d9
PubMed Abstract | CrossRef Full Text | Google Scholar
37. Detke MJ, Lu Y, Goldstein DJ, Hayes JR, Demitrack MA. Duloxetine, threescore mg once daily, for major depressive disorder: a randomized double-blind placebo-controlled trial. J Clin Psychiatry. (2002) 63:308–15. doi: x.4088/JCP.v63n0407
PubMed Abstract | CrossRef Total Text | Google Scholar
38. Goldstein DJ, Mallinckrodt C, Lu Y, Demitrack MA. Duloxetine in the treatment of major depressive disorder: a double-blind clinical trial. J Clin Psychiatry. (2002) 63:225–31. doi: 10.4088/JCP.v63n0309
CrossRef Full Text | Google Scholar
39. Burt VK, Wohlreich MM, Mallinckrodt CH, Detke MJ, Watkin JG, Stewart DE. Duloxetine for the treatment of major depressive disorder in women ages 40 to 55 years. Psychosomatics. (2005) 46:345–54. doi: ten.1176/appi.psy.46.iv.345
PubMed Abstract | CrossRef Full Text | Google Scholar
40. Perahia DG, Wang F, Mallinckrodt CH, Walker DJ, Detke MJ. Duloxetine in the handling of major depressive disorder: a placebo- and paroxetine-controlled trial. Eur Psychiatry. (2006) 21:367–78. doi: 10.1016/j.eurpsy.2006.03.004
PubMed Abstract | CrossRef Full Text | Google Scholar
41. Perahia DG, Gilaberte I, Wang F, Wiltse CG, Huckins SA, Clemens JW, et al. Duloxetine in the prevention of relapse of major depressive disorder: double-blind placebo-controlled study. Br J Psychiatry. (2006) 188:346–53. doi: x.1192/bjp.188.4.346
PubMed Abstract | CrossRef Full Text | Google Scholar
42. Perahia DGS, Pritchett YL, Kajdasz DK, Bauer M, Jain R, Russell JM, et al. A randomized, double-blind comparison of duloxetine and venlafaxine in the treatment of patients with major depressive disorder. J Psychiatr Res. (2008) 42:22–34. doi: 10.1016/j.jpsychires.2007.01.008
PubMed Abstract | CrossRef Full Text | Google Scholar
43. Perahia DGS, Quail D, Desaiah D, Corruble E, Fava M. Switching to duloxetine from selective serotonin reuptake inhibitor antidepressants: a multicenter trial comparing ii switching techniques. J Clin Psychiatry. (2008) 69:95–105. doi: 10.4088/JCP.v69n0113
PubMed Abstract | CrossRef Total Text | Google Scholar
44. Perahia DGS, Maina M, Thase ME, Spann ME, Wang F, Walker DJ, et al. Duloxetine in the prevention of depressive recurrences: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. (2009) 70:706–xvi. doi: 10.4088/JCP.08m04756
PubMed Abstract | CrossRef Full Text | Google Scholar
45. Fava M, Detke MJ, Balestrieri M, Wang F, Raskin J, Perahia D. Direction of low relapse: re-initiation of duloxetine handling or dose increase. J Psychiatr Res. (2006) 40:328–36. doi: 10.1016/j.jpsychires.2005.06.005
CrossRef Full Text | Google Scholar
46. Brecht Southward, Courtecuisse C, Debieuvre C, Croenlein J, Desaiah D, Raskin J, et al. Efficacy and safe of duloxetine 60 mg once daily in the treatment of pain in patients with major depressive disorder and at least moderate hurting of unknown etiology: a randomized controlled trial. J Clin Psychiatry. (2007) 68:1707–sixteen. doi: ten.4088/JCP.v68n1110
PubMed Abstract | CrossRef Full Text | Google Scholar
47. Lee P, Shu L, Xu X, Wang CY, Lee MS, Liu CY, et al. One time-daily duloxetine 60 mg in the treatment of major depressive disorder: multicenter, double-blind, randomized, paroxetine-controlled, non-inferiority trial in China, Korea, Taiwan and Brazil. Psychiatry Clin Neurosci. (2007) 61:295–307. doi: 10.1111/j.1440-1819.2007.01666.x
PubMed Abstract | CrossRef Full Text | Google Scholar
48. Brecht S, Desaiah D, Marechal ES, Santini AM, Podhorna J, Guelfi JD. Efficacy and rubber of duloxetine threescore mg and 120 mg daily in patients hospitalized for severe depression: a double-blind randomized trial. J Clin Psychiatry. (2011) 72:1086–94. doi: 10.4088/JCP.09m05723blu
PubMed Abstruse | CrossRef Full Text | Google Scholar
49. Pigott TA, Prakash A, Arnold LM, Aaronson ST, Mallinckrodt CH, Wohlreich MM. Duloxetine versus escitalopram and placebo: an eight-month, double-blind trial in patients with major depressive disorder. Curr Med Res Opin. (2007) 23:1303–xviii. doi: 10.1185/030079907X188107
PubMed Abstract | CrossRef Full Text | Google Scholar
50. Raskin J, Wiltse CG, Siegal A, Sheikh J, Xu J, Dinkel JJ, et al. Efficacy of duloxetine on cognition, depression, and pain in elderly patients with major depressive disorder: an 8-week, double-blind, placebo-controlled trial. Am J Psychiatry. (2007) 164:900–9. doi: 10.1176/ajp.2007.164.6.900
PubMed Abstract | CrossRef Full Text | Google Scholar
51. Raskin J, Wiltse CG, Dinkel JJ, Walker DJ, Desaiah D, Katona C. Safety and tolerability of duloxetine at 60 mg once daily in elderly patients with major depressive disorder. J Clin Psychopharmacol. (2008) 28:32–8. doi: 10.1097/jcp.0b013e318160738e
PubMed Abstruse | CrossRef Total Text | Google Scholar
52. Wise TN, Wiltse CG, Iosifescu DV, Sheridan M, Xu JY, Raskin J. The safety and tolerability of duloxetine in depressed elderly patients with and without medical comorbidity. Int J Clin Pract. (2007) 61:1283–93. doi: 10.1111/j.1742-1241.2007.01476.x
PubMed Abstract | CrossRef Full Text | Google Scholar
53. Karp JF, Whyte EM, Lenze EJ, Dew MA, Begley A, Miller Medico, et al. Rescue pharmacotherapy with duloxetine for selective serotonin reuptake inhibitor nonresponders in late-life low: outcome and tolerability. J Clin Psychiatry. (2008) 69:457–63. doi: 10.4088/JCP.v69n0317
PubMed Abstruse | CrossRef Total Text | Google Scholar
54. Kornstein SG, Dunner DL, Meyers AL, Whitmyer VG, Mallinckrodt CH, Wohlreich MM, et al. A randomized, double-bullheaded study of increasing or maintaining duloxetine dose in patients without remission of major depressive disorder after initial duloxetine therapy. J Clin Psychiatry. (2008) 69:1383–92. doi: x.4088/JCP.v69n0905
PubMed Abstract | CrossRef Full Text | Google Scholar
56. Herrera-Guzmán I, Herrera-Abarca JE, Gudayol-Ferré Due east, Herrera-Guzmán D, Gómez-Carbajal L, Peña-Olvira One thousand, et al. Effects of selective serotonin reuptake and dual serotonergic-noradrenergic reuptake treatments on attention and executive functions in patients with major depressive disorder. Psychiatry Res. (2010) 177:323–29. doi: 10.1016/j.psychres.2010.03.006
PubMed Abstract | CrossRef Full Text | Google Scholar
57. Volonteri LS, Colasanti A, Cerveri G, Fiorentini A, De Gaspari IF, Mauri MC, et al. Clinical outcome and tolerability of duloxetine in the treatment of major depressive disorder: a 12-week report with plasma levels. J Psychopharmacol. (2010) 24:1193–ix. doi: 10.1177/0269881109104863
PubMed Abstruse | CrossRef Full Text | Google Scholar
58. Sagman D, McIntosh D, Lee MS, Li H, Ruschel S, Hussain N, et al. Attributes of response in depressed patients switched to handling with duloxetine. Int J Clin Pract. (2011) 65:73–81. doi: 10.1111/j.1742-1241.2010.02546.10
PubMed Abstract | CrossRef Full Text | Google Scholar
59. Gaynor PJ, Gopal M, Zheng Due west, Martinez JM, Robinson MJ, Marangell LB. A randomized placebo-controlled trial of duloxetine in patients with major depressive disorder and associated painful concrete symptoms. Curr Med Res Opin. (2011) 27:1849–58. doi: x.1185/03007995.2011.609539
PubMed Abstract | CrossRef Full Text | Google Scholar
60. Martinez JM, Katon W, Greist JH, Kroenke K, Thase ME, Meyers AL, et al. A pragmatic 12-week, randomized trial of duloxetine versus generic selective serotonin-reuptake inhibitors in the treatment of adult outpatients in a moderate-to-astringent depressive episode. Int Clin Psychopharmacol. (2012) 27:17–26. doi: 10.1097/YIC.0b013e32834ce11b
PubMed Abstract | CrossRef Full Text | Google Scholar
61. Oakes TMM, Myers AL, Marangell LB, Ahl J, Prakash A, Thase ME, et al. Assessment of depressive symptoms and functional outcomes in patients with major depressive disorder treated with duloxetine versus placebo: primary outcomes from two trials conducted under the same protocol. Hum Psychopharmacol. (2012) 27:47–56. doi: x.1002/hup.1262
PubMed Abstract | CrossRef Full Text | Google Scholar
62. Rosso K, Rigardetto Due south, Bogetto F, Maina 1000. A randomized, unmarried-blind, comparison of duloxetine with bupropion in the treatment of SSRI-resistant major low. J Bear upon Disord. (2012) 136:172–half-dozen. doi: 10.1016/j.jad.2011.07.026
PubMed Abstract | CrossRef Total Text | Google Scholar
63. Robinson K, Oakes TM, Raskin JL, Liu P, Shoemaker S, Craig Nelson J. Acute and long-term handling of tardily-life major depressive disorder: duloxetine versus placebo. Am J Geriatr Psychiatry. (2014) 22:34–45. doi: x.1016/j.jagp.2013.01.019
PubMed Abstract | CrossRef Full Text | Google Scholar
64. Buoli Grand, Melter CC, Caldiroli A, Altamura AC. Are antidepressants equally effective in the long-term treatment of major depressive disorder? Hum Psychopharmacol. (2015) thirty:21–vii. doi: 10.1002/hup.2447
PubMed Abstract | CrossRef Full Text | Google Scholar
65. Mowla A, Dastgheib SA, Razeghian Jahromi L. Comparing the effects of sertraline with duloxetine for depression severity and symptoms: a double-blind, randomized controlled trial. Clin Drug Investig. (2016) 36:539–43. doi: 10.1007/s40261-016-0399-6
PubMed Abstract | CrossRef Full Text | Google Scholar
66. De Donatis D, Florio Five, Porcelli South, Saria A, Mercolini L, Serretti A, et al. Duloxetine plasma level and antidepressant response. Prog Neuro-Psychopharmacol Biol Psychiatry. (2019) 92:127–32. doi: x.1016/j.pnpbp.2019.01.001
PubMed Abstract | CrossRef Full Text | Google Scholar
67. Alaka KJ, Noble W, Montejo A, Dueñas H, Munshi A, Strawn JR, et al. Efficacy and safety of duloxetine in the treatment of older adult patients with generalized anxiety disorder: a randomized, double-blind, placebo-controlled trial. Int J Geriatr Psychiatry. (2014) 29:978–86. doi: 10.1002/gps.4088
PubMed Abstract | CrossRef Total Text | Google Scholar
68. Bodkin JA, Allgulander C, Llorca PM, Spann ME, Walker DJ, Russell JM, et al. Predictors of relapse in a written report of duloxetine treatment for patients with generalized anxiety disorder. Hum Psychopharmacol Clin Ext. (2011) 26:258–66. doi: 10.1002/hup.1211
PubMed Abstract | CrossRef Full Text | Google Scholar
69. Pierò A, Locati E. An open up, non-randomised comparison of escitalopram and duloxetine for the handling of subjects with generalized anxiety disorder. Hum Psychopharmacol Clin Ext. (2011) 26:63–71. doi: 10.1002/hup.1172
PubMed Abstruse | CrossRef Full Text | Google Scholar
70. Wu WY, Wang Yard, Brawl SG, Desaiah D, Ang QQ. Duloxetine versus placebo in the handling of patients with generalized anxiety disorder in China. Chin Med J. (2011) 124:3260–viii.
PubMed Abstract | Google Scholar
71. Nicolini H, Bakish D, Duenas H, Spann M, Erickson J, Hallberg C, et al. Improvement of psychic and somatic symptoms in adult patients with generalized feet disorder : examination from a duloxetine, venlafaxine extended-release and placebo-controlled trial. Psychol Med. (2009) 39:267–76. doi: 10.1017/S0033291708003401
PubMed Abstract | CrossRef Total Text | Google Scholar
72. Allgulander C, Nutt D, Detke M, Erickson J, Spann M, Walker D, et al. A not-inferiority comparison of duloxetine and venlafaxine in the treatment of adult patients with generalized anxiety disorder. J Psychopharmacol. (2008) 22:417–25. doi: 10.1177/0269881108091588
PubMed Abstract | CrossRef Total Text | Google Scholar
73. Davidson J, Allgulander C, Pollack MH, Hartford J, Erickson JS, Russell JM, et al. Efficacy and tolerability of duloxetine in elderly patients with generalized anxiety disorder: a pooled assay of iv randomized, double-blind, placebo-controlled studies. Hum Psychopharmacol. (2008) 23:519–26. doi: 10.1002/hup.949
PubMed Abstract | CrossRef Full Text | Google Scholar
74. Pollack MH, Kornstein SG, Spann ME, Crits-Christoph P, Raskin J, Russell JM. Early improvement during duloxetine handling of generalized anxiety disorder predicts response and remission at endpoint. J Psychiatr Res. (2008) 42:1176–84. doi: ten.1016/j.jpsychires.2008.02.002
PubMed Abstruse | CrossRef Total Text | Google Scholar
75. Russell JM, Weisberg R, Fava M, Hartford JT, Erickson JS, D'Souza DN. Efficacy of duloxetine in the handling of generalized anxiety disorder in patients with clinically significant pain symptoms. Depress Anxiety. (2008) 25:1–11. doi: x.1002/da.20337
PubMed Abstruse | CrossRef Total Text | Google Scholar
76. Rynn One thousand, Russell J, Erickson J, Detke MJ, Ball Due south, Dinkel J, et al. Efficacy and safety of duloxetine in the handling of generalized anxiety disorder: a flexible-dose, progressive-titration, placebo-controlled trial. Depress Anxiety. (2008) 25:182–9. doi: x.1002/da.20271
PubMed Abstract | CrossRef Full Text | Google Scholar
77. Hartford J, Kornstein S, Liebowitz Yard, Pigott T, Russell J, Detke M, et al. Duloxetine every bit an SNRI treatment for generalized anxiety disorder : results from a placebo and active-controlled trial. Int Clin Psychopharmacol. (2007) 22:167–74. doi: 10.1097/YIC.0b013e32807fb1b2
PubMed Abstract | CrossRef Full Text | Google Scholar
78. Salehifar Eastward, Janbabaei G, Hendouei Northward, Alipour A, Tabrizi N, Avan R. Comparison of the efficacy and safety of pregabalin and duloxetine in taxane-induced sensory neuropathy: a randomized controlled trial. Clin Drug Investig. (2020) 40:249–57. doi: ten.1007/s40261-019-00882-6
PubMed Abstract | CrossRef Full Text | Google Scholar
79. Jha Southward, Sahani OP, Siddiqui S, Verma MK, Mazumder A, Waghdhare S. Effectiveness of pregabalin compared to duloxetine in diabetic peripheral neuropathic pain: an observational study. J Assoc Physicians India. (2019) 67:32–6.
PubMed Abstruse | Google Scholar
lxxx. Farshchian N, Alavi A, Heydarheydari S, Moradian N. Comparative study of the effects of venlafaxine and duloxetine on chemotherapy-induced peripheral neuropathy. Cancer Chemother Pharmacol. (2018) 82:787–93. doi: 10.1007/s00280-018-3664-y
PubMed Abstract | CrossRef Total Text | Google Scholar
81. Schukro RP, Oehmke MJO, Geroldinger A, Heinze G, Kress H, Pramhas Due south. Efficacy of duloxetine in chronic depression back pain with a neuropathic component a randomized, double-blind, placebo-controlled crossover trial. Anesthesiology. (2016) 124:150–8. doi: 10.1097/ALN.0000000000000902
PubMed Abstruse | CrossRef Full Text | Google Scholar
82. Yasuda H, Hotta North, Kasuga K, Kashiwagi A, Kawamori R, Yamada T, et al. Efficacy and condom of 40 mg or lx mg duloxetine in Japanese adults with diabetic neuropathic hurting: results from a randomized, 52-calendar week, open-label report. J Diabetes Investig. (2016) 7:100–8. doi: ten.1111/jdi.12361
PubMed Abstruse | CrossRef Full Text | Google Scholar
83. Gao Y, Guo X, Han P, Li Q, Yang G, Qu S, et al. Treatment of patients with diabetic peripheral neuropathic pain in Cathay: a double-blind randomised trial of duloxetine vs. placebo. Int J Clin Pract. (2015) 69:957–66. doi: x.1111/ijcp.12641
PubMed Abstruse | CrossRef Full Text | Google Scholar
84. Happich 1000, Schneider E, Boess FG, Wilhelm Southward, Schacht A, Birklein F, et al. Effectiveness of duloxetine compared with pregabalin and gabapentin in diabetic peripheral neuropathic hurting results from a german observational report. Clin J Hurting. (2014) 30:875–85. doi: ten.1097/AJP.0000000000000057
PubMed Abstract | CrossRef Full Text | Google Scholar
85. Irving G, Tanenberg RJ, Raskin J, Risser RC, Malcolm S. Comparative safety and tolerability of duloxetine vs. pregabalin vs duloxetine plus gabapentin in patients with diabetic peripheral neuropathic pain. Int J Clin Pract. (2014) 68:1130–twoscore. doi: 10.1111/ijcp.12452
PubMed Abstract | CrossRef Full Text | Google Scholar
86. Vollmer TL, Robinson MJ, Risser RC, Malcolm SK. A randomized, double-blind, placebo-controlled trial of duloxetine for the treatment of pain in patients with multiple sclerosis. Hurting Pract. (2014) 14:732–44. doi: x.1111/papr.12127
PubMed Abstruse | CrossRef Full Text | Google Scholar
87. Smith EML, Pang H, Cirrincione C, Felishman Due south, Paskett ED, Bressler LR, et al. Effect of duloxetine on pain, function, and quality of life amid patients with chemotherapy-induced painful peripheral neuropathy: a randomized clinical trial. JAMA. (2013) 309:1359–67. doi: 10.1001/jama.2013.2813
PubMed Abstruse | CrossRef Full Text | Google Scholar
88. Tesfaye South, Wilhelm S, Lledo A, Schacht A, Tölle T, Bouhassira D, et al. Duloxetine and pregabalin: loftier-dose monotherapy or their combination? the "philharmonic-DN study" - a multinational, randomized, double-blind, parallel-grouping study in patients with diabetic peripheral neuropathic hurting. Pain. (2013) 154:2616–25. doi: 10.1016/j.pain.2013.05.043
PubMed Abstract | CrossRef Full Text | Google Scholar
89. Boyle J, Eriksson MEV, Gribble L, Gouni R, Johnsen S, Coppini DV, et al. Randomized, placebo-controlled comparison of amitriptyline, duloxetine, and pregabalin in patients with chronic diabetic peripheral neuropathic pain: impact on pain, polysomnographic sleep, daytime performance, and quality of life. Diabetes Care. (2012) 35:2451–8. doi: 10.2337/dc12-0656
PubMed Abstruse | CrossRef Full Text | Google Scholar
90. Tanenberg RJ, Irving GA, Risser RC, Ahl J, Robinson MJ, Skljarevski 5, et al. Duloxetine, pregabalin, and duloxetine plus gabapentin for diabetic peripheral neuropathic pain management in patients with inadequate pain response to gabapentin: an open up-label, randomized, noninferiority comparison. Mayo Clin Proc. (2011) 86:615–26. doi: 10.4065/mcp.2010.0681
PubMed Abstract | CrossRef Total Text | Google Scholar
91. Skljarevski V, Desaiah D, Chappell AS, Detke MJ, Gross JL, Ziegler D. Evaluating the maintenance of consequence of duloxetine in patients with diabetic peripheral neuropathic pain. Diabetes Metab Res Rev. (2009) 25:623–31. doi: 10.1002/dmrr.1000
PubMed Abstract | CrossRef Full Text | Google Scholar
92. Armstrong DG, Chappell Equally, Le TK, Kajdasz DK, Backonja K, D'Souza DN, et al. Duloxetine for the management of diabetic peripheral neuropathic pain: Evaluation of functional outcomes. Pain Med. (2007) 8:410–8. doi: ten.1111/j.1526-4637.2007.00276.x
PubMed Abstract | CrossRef Full Text | Google Scholar
93. Wernicke JF, Wang F, Pritchett YL, Smith TR, Raskin J, Souza DND, et al. An open up-label 52-week clinical extension comparison duloxetine with routine care in patients with diabetic. Pain Med. (2007) viii:503–13. doi: 10.1111/j.1526-4637.2006.00258.ten
PubMed Abstract | CrossRef Full Text | Google Scholar
94. Raskin J, Wang F, Pritchett YL, Goldstein DJ. Duloxetine for patients with diabetic peripheral neuropathic hurting: a half dozen-month open-label safety report. Pain Med. (2006) 7:373–85. doi: 10.1111/j.1526-4637.2006.00207.x
PubMed Abstract | CrossRef Full Text | Google Scholar
96. Raskin J, Pritchett YL, Wang F, D'Souza DN, Waninger AL, Iyengar Due south, et al. A double-blind, randomized multicenter trial comparing duloxetine with placebo in the management of diabetic peripheral neuropathic pain. Pain Med. (2005) 6:346–56. doi: 10.1111/j.1526-4637.2005.00061.ten
PubMed Abstruse | CrossRef Full Text | Google Scholar
97. Murakami Chiliad, Osada K, Ichibayashi H, Mizuno H, Ochiai T, Ishida Yard, et al. An open-label, long-term, stage III extension trial of duloxetine in Japanese patients with fibromyalgia. Mod Rheumatol. (2017) 27:688–95. doi: ten.1080/14397595.2016.1245237
PubMed Abstract | CrossRef Full Text | Google Scholar
98. Murakami M, Osada K, Mizuno H, Ochiai T, Alev L, Nishioka K. A randomized, double-bullheaded, placebo-controlled phase III trial of duloxetine in Japanese fibromyalgia patients. Arthritis Res Ther. (2015) 17:1–13. doi: 10.1186/s13075-015-0718-y
PubMed Abstract | CrossRef Total Text | Google Scholar
99. Mohs R, Mease P, Arnold LM, Wang F, Ahl J, Gaynor PJ, et al. The effect of duloxetine treatment on cognition in patients with fibromyalgia. Psychosom Med. (2012) 74:628–34. doi: 10.1097/PSY.0b013e31825b9855
PubMed Abstract | CrossRef Full Text | Google Scholar
100. Arnold LM, Hudson JI, Wang F, Wohlreich MM, Prakash A, Kajdasz DK, et al. Comparisons of the efficacy and safe of duloxetine for the treatment of fibromyalgia in patients with versus without major depressive disorder. Clin J Pain. (2009) 25:461–eight. doi: ten.1097/AJP.0b013e318197d4e4
PubMed Abstract | CrossRef Full Text | Google Scholar
101. Chappell AS, Littlejohn M, Kajdasz DK, Scheinberg Grand, D'souza DN, Moldofsky H. A 1-year condom and efficacy study of duloxetine in patients with fibromyalgia. Clin. J. Pain. (2009) 25:365–75. doi: 10.1097/AJP.0b013e31819be587
PubMed Abstract | CrossRef Total Text | Google Scholar
102. Russell IJ, Mease PJ, Smith TR, Kajdasz DK, Wohlreich MM, Detke MJ, et al. Efficacy and safety of duloxetine for treatment of fibromyalgia in patients with or without major depressive disorder: results from a six-month, randomized, double-blind, placebo-controlled, stock-still-dose trial. Pain. (2008) 136:432–44. doi: x.1016/j.pain.2008.02.024
PubMed Abstract | CrossRef Total Text | Google Scholar
103. Arnold LM, Rosen A, Pritchett YL, D'Souza DN, Goldstein DJ, Iyengar Due south, et al. A randomized, double-blind, placebo-controlled trial of duloxetine in the treatment of women with fibromyalgia with or without major depressive disorder. Pain. (2005) 119:5–15. doi: 10.1016/j.pain.2005.06.031
CrossRef Full Text | Google Scholar
104. Arnold LM, Lu Y, Crofford LJ, Wohlreich M, Detke MJ, Iyengar S, et al. A double-blind, multicenter trial comparing duloxetine with placebo in the treatment of fibromyalgia patients with or without major depressive disorder. Arthritis Rheum. (2004) 50:2974–84. doi: 10.1002/fine art.20485
CrossRef Total Text | Google Scholar
105. Cornu JN, Merlet B, Ciofu C, Mouly S, Peyrat L, Sbe P, et al. Duloxetine for mild to moderate postprostatectomy incontinence: Preliminary results of a randomised, placebo-controlled trial. Eur Urol. (2011) 59:148–54. doi: ten.1016/j.eururo.2010.10.031
PubMed Abstruse | CrossRef Full Text | Google Scholar
106. Cardozo Fifty, Lange R, Voss S, Beardsworth A, Manning M, Viktrup L, et al. Curt-and long-term efficacy and safety of duloxetine in women with predominant stress urinary incontinence. Curr Med Res Opin. (2010) 26:253–61. doi: x.1185/03007990903438295
PubMed Abstruse | CrossRef Full Text | Google Scholar
107. Bent AE, Gousse AE, Hendrix SL, Klutke CG, Monga AK, Yuen CK, et al. Duloxetine compared with placebo for the handling of women with mixed urinary incontinence. Neurourol Urodyn. (2008) 27:212–21. doi: 10.1002/nau.20471
PubMed Abstract | CrossRef Full Text | Google Scholar
108. Lin ATL, Sun MJ, Tai HL, Chuang YC, Huang ST, Wang North, et al. Duloxetine versus placebo for the treatment of women with stress predominant urinary incontinence in Taiwan: a double-blind, randomized, placebo-controlled trial. BMC Urol. (2008) viii:1–9. doi: 10.1186/1471-2490-8-ii
PubMed Abstract | CrossRef Full Text | Google Scholar
109. Schagen van Leeuwen JH, Lange RR, Jonasson AF, Chen WJ, Viktrup L. Efficacy and safety of duloxetine in elderly women with stress urinary incontinence or stress-predominant mixed urinary incontinence. Maturitas. (2008) 60:138–47. doi: 10.1016/j.maturitas.2008.04.012
CrossRef Total Text | Google Scholar
110. Castro-Diaz D, Palma PCR, Bouchard C, Haab F, Hampel C, Carone R, et al. Effect of dose escalation on the tolerability and efficacy of duloxetine in the treatment of women with stress urinary incontinence. Int Urogynecol J. (2007) 18:919–29. doi: 10.1007/s00192-006-0256-x
PubMed Abstract | CrossRef Full Text | Google Scholar
111. Schlenker B, Gratzke C, Reich O, Schorsch I, Seitz M, Stief CG. Preliminary results on the off-label apply of duloxetine for the handling of stress incontinence afterwards radical prostatectomy or cystectomy. Eur Urol. (2006) 49:1075–eight. doi: 10.1016/j.eururo.2006.01.038
CrossRef Full Text | Google Scholar
112. Weinstein DL, Cohen JS, Liu C, Meadows ES, Plouffe L, Muram D. Duloxetine in the treatment of women with stress urinary incontinence: results from DESIRE (Duloxetine Efficacy and Safety for Incontinence in Racial and Ethnic populations). Curr Med Res Opin. (2006) 22:2121–9. doi: ten.1185/030079906X148337
PubMed Abstract | CrossRef Full Text | Google Scholar
113. Ghoniem GM, Van Leeuwen JS, Elser DM, Freeman RM, Zhao YD, Yalcin I, et al. A randomized controlled trial of duloxetine alone, pelvic floor muscle training alone, combined treatment and no active treatment in women with stress urinary incontinence. J Urol. (2005) 173:1647–53. doi: ten.1097/01.ju.0000154167.90600.c6
PubMed Abstruse | CrossRef Full Text | Google Scholar
114. Kinchen KS, Obenchain R, Swindle R. Impact of duloxetine on quality of life for women with symptoms of urinary incontinence. Int Urogynecol J. (2005) 16:337–44. doi: 10.1007/s00192-004-1270-5
PubMed Abstract | CrossRef Total Text | Google Scholar
115. Cardozo Fifty, Drutz HP, Baygani SK, Crash-land RC. Pharmacological treatment of women awaiting surgery for stress urinary incontinence. Obstet Gynecol. (2004) 104:511–9. doi: 10.1097/01.AOG.0000134525.86480.0f
PubMed Abstract | CrossRef Total Text | Google Scholar
116. Millard RJ, Moore K, Rencken R, Yalcin I, Bump RC. Duloxetine vs placebo in the treatment of stress urinary incontinence: a four-continent randomized clinical trial. BJU Int. (2004) 93:311–viii. doi: x.1111/j.1464-410X.2004.04607.x
PubMed Abstract | CrossRef Total Text | Google Scholar
117. Van Kerrebroeck P, Abrams P, Lange R, Slack M, Wyndaele JJ, Yalcin I, et al. Duloxetine versus placebo in the treatment of European and Canadian women with stress urinary incontinence. BJOG An Int J Obstet Gynaecol. (2004) 111:249–57. doi: 10.1111/j.1471-0528.2004.00067.ten
PubMed Abstract | CrossRef Full Text | Google Scholar
118. Dmochowski RR, Miklos JR, Norton PA, Zinner NR, Yalcin I, Bump RC. Duloxetine versus placebo for the treatment of North American women with stress urinary incontinence. J Urol. (2003) 170:1259–63. doi: x.1097/01.ju.0000080708.87092.cc
PubMed Abstract | CrossRef Full Text | Google Scholar
119. Bradley LA, Wohlreich MM, Wang F, Gaynor PJ, Robinson MJ, D'Souza DN, et al. Pain response profile of patients with fibromyalgia treated with duloxetine. Clin J Pain. (2010) 26:498–504. doi: 10.1097/AJP.0b013e3181dee80e
PubMed Abstruse | CrossRef Total Text | Google Scholar
120. Moher D, Liberati A, Tetzlaff J, Altman D. Preferred reporting items for systematic reviews and metaanalyses: the PRISMA Statement. Ann Intern Med. (2009) 151:264–9. doi: 10.7326/0003-4819-151-iv-200908180-00135
CrossRef Full Text | Google Scholar
121. Munn Z, Moola Southward, Riitano D, Lisy K. The evolution of a critical appraisal tool for use in systematic reviews addressing questions of prevalence. Int J Heal Policy Manag. (2014) 3:123–viii. doi: ten.15171/ijhpm.2014.71
PubMed Abstract | CrossRef Total Text | Google Scholar
123. Naranjo C, Del Reguero Fifty, Moratalla Yard, Hercberg Yard, Valenzuela K, Failde I. Anxiety, low and sleep disorders in patients with diabetic neuropathic pain: a systematic review. Expert Rev Neurother. (2019) 19:1201–9. doi: x.1080/14737175.2019.1653760
PubMed Abstract | CrossRef Full Text | Google Scholar
124. Vileikyte L, Leventhal H, Gonzalez JS, Peyrot G, Rubin RR, Ulbrecht JS, et al. Diabetic peripheral neuropathy and depressive symptoms: the association revisited. Diabetes Intendance. (2005) 28:2378–83. doi: ten.2337/diacare.28.ten.2378
PubMed Abstruse | CrossRef Total Text | Google Scholar
125. Arnold LM, Hudson JI, Keck PE, Auchenbach MB, Javaras KN, Hess EV. Comorbidity of fibromyalgia and psychiatric disorders. J Clin Psychiatry. (2006) 67:1219–25. doi: 10.4088/JCP.v67n0807
PubMed Abstract | CrossRef Full Text | Google Scholar
126. Heymen S. Psychological and cognitive variables affecting treatment outcomes for urinary and fecal incontinence. Gastroenterology. (2004) 126:S146–51. doi: 10.1053/j.gastro.2003.ten.040
PubMed Abstract | CrossRef Full Text | Google Scholar
128. Haase J, Brown E. Integrating the monoamine, neurotrophin and cytokine hypotheses of depression - a fundamental office for the serotonin transporter? Pharmacol Ther. (2015) 147:1–11. doi: x.1016/j.pharmthera.2014.10.002
PubMed Abstract | CrossRef Total Text | Google Scholar
129. Montoya A, Bruins R, Katzman MA, Blier P. The noradrenergic paradox: implications in the direction of depression and anxiety. Neuropsychiatr Dis Treat. (2016) 12:541–57. doi: 10.2147/NDT.S91311
PubMed Abstract | CrossRef Total Text | Google Scholar
130. Liu Y, Zhao J, Fan X, Guo W. Dysfunction in serotonergic and noradrenergic systems and somatic symptoms in psychiatric disorders. Front end Psychiatry. (2019) 10:286. doi: ten.3389/fpsyt.2019.00286
PubMed Abstruse | CrossRef Full Text | Google Scholar
131. Nekovarova T, Yamamotova A, Vales M, Stuchlik A, Fricova J, Rokyta R. Mutual mechanisms of pain and depression: are antidepressants besides analgesics? Front Behav Neurosci. (2014) viii:99. doi: 10.3389/fnbeh.2014.00099
PubMed Abstract | CrossRef Total Text | Google Scholar
132. Miyazato M, Kaiho Y, Kamo I, Chancellor MB, Sugaya Thousand, De Groat WC, et al. Effect of duloxetine, a norepinephrine and serotonin reuptake inhibitor, on sneeze-induced urethral continence reflex in rats. Am J Physiol - Ren Physiol. (2008) 295:F264–71. doi: 10.1152/ajprenal.90241.2008
PubMed Abstract | CrossRef Full Text | Google Scholar
133. Sultan A, Gaskell H, Derry South, Andrew RA. Duloxetine for painful diabetic neuropathy and fibromyalgia pain: systematic review of randomised trials. BMC Neurol. (2008) 8:29. doi: x.1186/1471-2377-8-29
PubMed Abstract | CrossRef Full Text | Google Scholar
135. Lisinski A, Hieronymus F, Näslund J, Nilsson Southward, Eriksson East. Detail-based analysis of the furnishings of duloxetine in low: a patient-level post hoc report. Neuropsychopharmacology. (2019) 45:553–sixty. doi: 10.1038/s41386-019-0523-4
PubMed Abstruse | CrossRef Full Text | Google Scholar
136. Sharma A, Goldberg MJ, Cerimele BJ. Pharmacokinetics and condom of duloxetine, a dual-serotonin and norepinephrine reuptake inhibitor. J Clin Pharmacol. (2000) 40:161–7. doi: 10.1177/00912700022008810
PubMed Abstract | CrossRef Full Text | Google Scholar
138. De Heer EW, Gerrits MMJG, Beekman ATF, Dekker J, Van Marwijk HWJ, De Waal MWM, et al. The association of low and anxiety with pain: a report from NESDA. PLoS ONE. (2014) ix:e106907. doi: 10.1371/periodical.pone.0106907
PubMed Abstruse | CrossRef Full Text | Google Scholar
140. Vinader-Caerols C, Monleón Due south, Carrasco C, Parra A. Effects of alcohol, coffee, and tobacco, alone or in combination, on physiological parameters and anxiety in a immature population. J Caffeine Res. (2012) 2:seventy–half dozen. doi: 10.1089/jcr.2012.0018
PubMed Abstract | CrossRef Full Text | Google Scholar
141. Fric M, Pfuhlmann B, Laux G, Riederer P, Distler 1000, Artmann S, et al. The influence of smoking on the serum level of duloxetine. Pharmacopsychiatry. (2008) 41:151–5. doi: 10.1055/s-2008-1073173
PubMed Abstract | CrossRef Full Text | Google Scholar
142. Vuppalanchi R, Hayashi PH, Chalasani North, Fontana RJ, Bonkovsky H, Saxena R, et al. Duloxetine hepatotoxicity: a case-series from the drug-induced liver injury network. Aliment Pharmacol Ther. (2010) 32:1174–83. doi: 10.1111/j.1365-2036.2010.04449.ten
PubMed Abstract | CrossRef Full Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fpsyt.2020.554899/full
Enregistrer un commentaire for "Treatment of Neuropathic Pain With Venlafaxine a Systematic Review"